Have a question ?
08045479695- Home Page
- Company Profile
-
Our Products
- CRITICAL CARE INJECTION
- ANTI CANCER MEDICINES
- LENALIDOMIDE CAPSULES 25MG
- LENALIDOMIDE CAPSULES 10MG
- Bretolap Tablets
- THALIDOMIDE CAPSULES 50MG
- GEMCITABINE 1000MG INJECTION
- Exemestane 25mg Tablet
- TEMOZOLOMIDE CAPSULES 20MG
- Ibrutinib Capsules 140mg
- Rucaparib Tablets
- L-ASPARIGENASE INJECTION 5000 IU
- IFOSFAMIDE 2000MG INJECTION WITH MESNA
- TEMOZOLAMIDE CAPSULES 100MG
- CARBOPLATIN 150MG INJECTION
- CISPLATIN 50MG INJECTION
- Letrozole Tab
- METHOTREXATE TABLETS 2.5MG
- CAPECITABINE TABLETS CAP 500MG
- Abiraterone Acetate
- TEMOZOLAMIDE CAPSULES 250MG
- BLEOMYCIN 15IU INJECTION
- CYTARABINE 1000MG INJECTION
- DACARBAZINE 100MG INJECTION
- DACARBAZINE 200MG INJECTION
- TEMOZOLOMIDE CAPSULES 250MG
- TEMOZOLOMIDE CAPSULES 100MG
- DOCETAXEL 120MG INJECTION
- IRINOTECAN 100MG INJECTION
- HYDROXYUREA CAPSULES IP 500MG
- MEGESTROL ACETATE TABLETS 160MG
- MEGESTROL ACETATE TAB
- MEGASTROL TABLETS 40MG
- L-ASPARIGENASE INJECTION 10000 IU
- METHOTREXATE 50MG INJECTION
- OXALIPLATIN 100MG INJECTION
- Pemetrexed 500mg Injection
- PROCARBAZINE HCL CAPSULES
- ANASTRAZOLE TABLETS 1MG
- TACROLIMUS CAPSULES 2MG
- IFOSFAMIDE 1000MG INJECTION WITH MESNA
- CYCLOPHOSPHAMIDE TABLETS 50MG
- TACROLIMUS CAPSULES 1MG
- Imatinib Mesylate Capsules 100mg
- PLERIXAFOR INJECTION
- TAMOXIFEN TABLETS 20MG
- DACTINOMYCIN 0.5MG INJECTION
- AFATINIB TABLETS
- ACALABRUTINIB CAPSULES
- ELTROMBOPAG OLAMINE TABLETS
- LENVATINIB CAPSULE
- BUSULFAN TABLETS 2MG
- 5 FLUROURACIL 500MG INJECTION
- EXEMESTANE TABLETS 25MG
- AZACITADINE 100MG
- CARMUSTIN 100MG INJECTION
- CYCLOPHOSPHAMIDE 1GM INJECTION
- CYCLOPHOSPHAMIDE 500MG INJECTION
- DACITABINE INJECTION 50MG
- Axitinib Tablets 5mg
- Erlotinib Tablets 150mg
- OSIMERTINIB TABLETS
- Chlorambucil Tablets 2mg
- ANASTRAZOLE TABLETS
- NIRAPARIB CAPSULES
- AZACITIDINE TABLETS
- Pomalidomide Capsule
- FOSAPREPITANT 150MG INJECTION
- CABOZANTINIB TABLET
- DASATINIB TAB 50MG
- Olaparib Capsules
- Nilotinib Capsules
- Estramustine Phosphate Capsule
- Pazopanib Tablets
- Sunitinib Capsule
- Flutamide Tablet
- Apalutamide Tablet
- Tucatinib Tablets
- Everolimus 10mg Tablets
- Temozolomide Capsules 250mg
- Lenalidomide 5mg Capsules
- Abiraterone Acetate Tablets
- Bicalutamide 50mg Tablet
- Granitero- Granisetron Inejction
- 40mg Lomustine Capsules
- Thiotepa For Injection 100mg
- Methocen 5
- MEGASTROL TABLETS 160MG
- MELPHALAN 50MG INJECTION
- Imaticen 400
- EPIRUBICIN 50MG INJECTION
- PACLITAXEL 300MG INJECTION
- Exemestane Tablets
- Imatinib Mesylate Tablets IP -400mg
- Mercaptopurine 50mg Tablets
- Megestrol 40mg Tablets
- REGORAFENIB TABLETS 40MG
- LAPATINIB TABLETS 250MG
- SORAFENIB TABLETS 200MG
- TRETINOIN SOFT GELATIN CAPSULES
- Bevacizumab INJECTION
- MELPHALAN TABLETS 2MG
- Anastrozole Tablet IP 1mg
- Etoposide Capsules 50mg
- Thioguanine Tablets
- Calcium Folinate Tablets 15mg
- Gefitinib Tablets 250mg
- Leucovorin Calcium 15mg Tablets
- ANTI HIV MEDICINE
- HEPATITS C MEDICINES
- PHARMACEUTICAL PRODUCTS
- Folic Acid 400mcg Tablets
- Curefast-o Syrup
- Nimodipine 30mg Tablet
- Iodixanol Injection 320mg
- Nitrofurantoin Capsules
- Paracetamol, Ibuprofen,CHlopheniramine, Ascorbic Acid, Phenylephrine HCL & Caffeine Sachets
- Aceclofenac, Paracetamol & Serratiopeptidase Tablets
- ACECLOFENAC 100MG and PARACETAMOL 500MG TABLETS
- SALBUTAMOL NEBULISER SOLUTION
- Vitamins B1, B6, B12 Tablets
- Thiamine Mononitrate, Pyridoxine Hydrochloride, Cyanocobalamin Tablets
- Haemorrhoidal Suppositories
- Lercanidipine 10mg Tablet
- CHLORPROMAZINE HYDROCHLORIDE 100MG TABLET
- ferrous sulphate 150mg + Folic acid 5mg capsules
- IBUPROFEN SOFTGEL CAPSULE
- Paracetamol with Ibuprofen Tablets
- Norethisterone Tablet
- Ocezole 20 - Omeprazole Capsules 20mg
- Bacillus Clausii spores suspension
- ANTI CANCER INJECTION
- Docetaxel 80mg Injection
- Iron Sucrose Injection 100mg
- LEUCOVORIN CALCIUM 50MG INJECTION
- ETOPOSIDE 100MG INJECTION
- DOXORUBICIN INJECTION
- Zoledronic Acid 4mg Injection
- VINCRISTINE 1MG Injection
- L-ASPARAGINASE FOR INJECTION
- Carboplatin Injection
- DAUNORUBICIN INJECTION
- RITUXIMAB CONCENTRATE FOR SOLUTION FOR INFUSION 10MG/ML
- OCTREOTIDE LAR INJECTION
- ARSENIC TRIOXIDE INJECTION 1MG/ML
- FULVESTRANT INJECTION 250MG 5ML
- Omeprazole for Injection
- Fludarabine Phosphate Injection
- Cladribine Injection
- Carfilzomib Injection
- Topotecan Hydrochloride Injection
- Vinorelbine Injection
- Granisetron Injection
- Decitabine 50mg Injection
- Degarelix Injection
- Peg Filgrastim Injection
- GOSERELIN INJECTION
- Filgrastim Injection Pfs 300mcg
- Trastuzumab For Injection
- Amifostine Injection
- IDARUBICIN INJECTION
- Azacitidine injection 100mg
- Fluorouracil injection
- Carboplatin Injection
- Pemetrexed Injection 500mg
- Nimotuzab Injection
- Leuprolide Acetate For Injection 11.25mg
- Busulfan Injection 60mg10ml
- Peg L-asparaginase Injection
- Vinblastine Sulphate Injection
- Mitoxantrone Injection 20mg
- Mitomycin For Injection
- Bacteriostatic Water 10ml
- Adalimumab Injection
- Cabazitaxel Injection 60mg/1.5ml
- Clofarabine Injection 20mg/20ml
- Bendamustine Hydrochloride For Injection
- Anti d Immunoglobulin Injection
- Paclitaxel (Protein-bound Particle) For Injectable Suspension
- Pembrolizumab Injection
- PHARMACEUTICAL OINMENTS AND CREAM -SKIN CARE
- EYE & EAR DROPS
- Cyclomune Eye Drops
- ATROPINE EYE DROPS
- Oflox Eye/ear Drops
- BIMATOPROST OPHTHALMIC SOLUTION
- TIMOLOL MALEATE EYE DROPS
- Azelastine Hydrochloride Eye Drops
- NAPHAZOLINE HCI, CHLORPHENIRAMINE MALEATE, PHENYLEPHRINE HCI, MENTHOL & CAMPHOR EYE DROPS
- CYCLOSIM EYE DROPS 0.1%
- FLUCONAZOLE EYE/EAR DROPS
- BECLOMETHASONE DIPROPIONATE, NEOMYCIN & CLOTRIMAZOLE EAR DROPS
- CARBOXYMETHYLCELLULOSE EYE DROPS 1.0% W/V
- PHENYLEPHERIN HCL 10% drops
- CYCLOSPORIN EYE DROPS
- TROPICAMIDE C PHENYLEPHRINE EYE DROP
- Brimonidine Tartrate and Timolol Maleate Eye drop
- CARBOXY-METHYCELLULOSE SODIUM EYE DROPS
- TRAVOPROST EYE DROPS
- NEPAFENAC OPHTHALMIC SUSPENSION
- MOXIFLOXACIN DEXAMETHASONE EYE DROPS
- TOBRAMYCIN OPHTHALMIC SOLUTION
- BRIMONIDINE TARTRATE TIMOLOL MALEATE EYE DROPS
- HYDROXYPROPYL METHYLCELLULOSE EYE DROPS
- Prednisolone Acetate Ophthalmic Suspension
- CARMELLOSE SODIUM EYE DROPS
- Latanoprost Ophthalmic Solution
- Dorzolamide Timolol Eye Drops
- Travoprost Timolol Ophthalmic Solution
- Sodium Chloride Ophthalmic Solution
- Tropicamide Eye Drops
- Dorzolamide With Timolol Ophthalmic Solution Usp
- Dorzolamide Eye Drops 2% W/v
- PHARMACEUTICAL INJECTION
- Lopamidol Injection
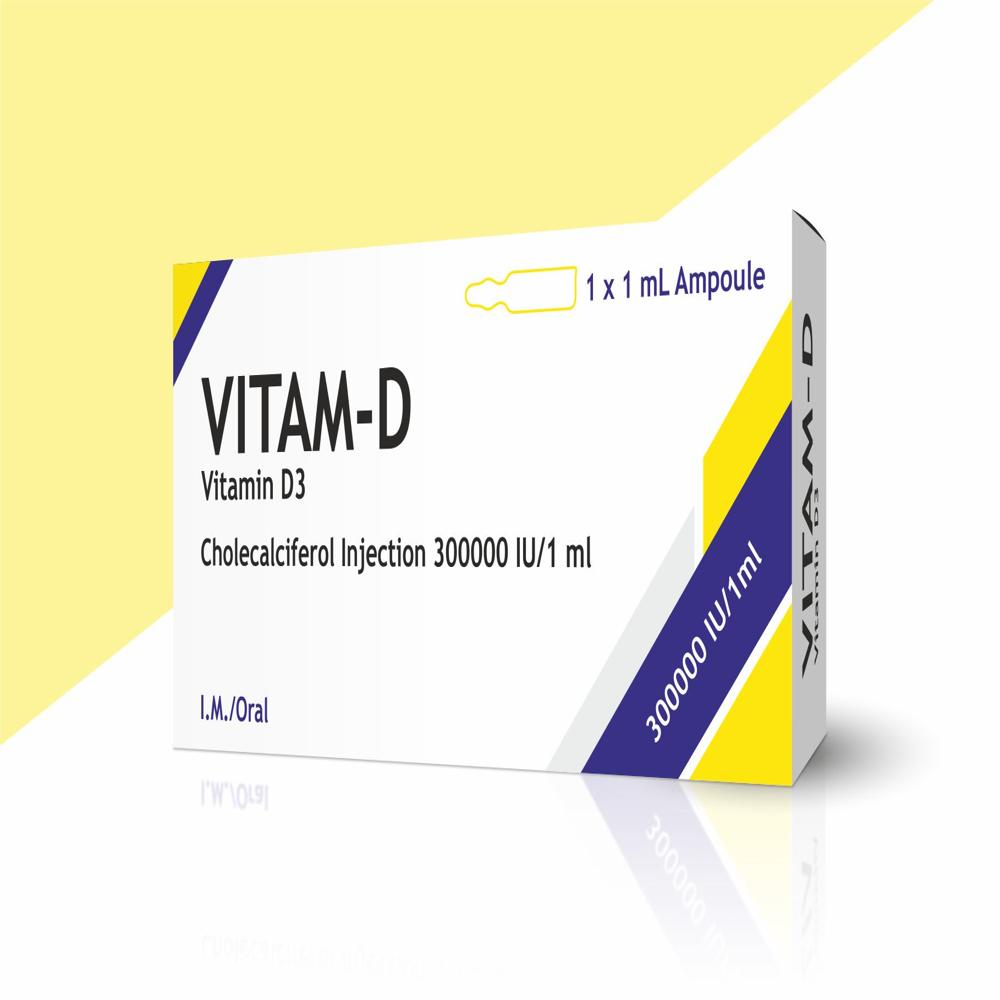
- VITAMIN D3 INJECTION
- Zinc chloride injection
- Vitamin H 5mg
- Progesterone Injection
- Sterile Co Trimoxazole Solution For Infusion
- Atropine Sulphate Injection 0.6MG/1ML
- Polidocanol injection 60mg/3ml
- Diatrizoate Meglumine And Diatrizoate Sodium Injection 76 100ml
- Betamethasone Sodium Phosphate Injection 4mg/2ml
- Iohexol Injection 300
- Clonidine HCL Injection 150mcg
- Furosemide Injection 20mg /2ml
- Bacteriostatic Water 5ml
- Nicorandil Injection 48mg
- Diclofenac Sodium Injection 75mg /3ml
- Ethamsylate injection 250mg / 2ml
- Adenosine injection 6mg /2ml
- Clemastine Injection 2mg/2ml
- Gadopentetate Dimeglumine Injection 20ml
- Glycopyrrolate Injection
- Lignocaine HCL injection 2% 30ml
- Oxytocin injection 10IU/ml
- Pantoprazole Injection 40mg
- Edaravone Injection 30mg/20ml
- Sodium Tetradecyl Sulphate Injection 60mg/2ml
- L-ornithine L-aspartate Infusion Concentrate
- Esmolol Injection 100mg/10ml
- Lignocaine injection 2% 2ml
- ISOFLURANE 100ML
- Glyceryl trinitrate injection 50mg/10ml
- Thiocolchicoside injection 4mg /2ml
- Cimetidine Injection 200mg
- Ondansetron Hydrochloride Injection 8mg/4ml
- Famotidine injection 20mg /2ml
- Chloropyramine 20mg/2ml Injection
- Thioctic acid injection 150mg/6ml
- Methylprednisolone Acetate Injection 80mg/2ml
- Diclofenac Sodium + Dicyclomine injection
- Sterile Noradernaline Concerate Ip
- Potassium Chloride Injection 150mg/ml
- Bupivacaine Hydrochloride Injection 0.5% 20ml
- Iodixanol injection 320mg
- Ibandronic Acide injection 6mg
- Ketorolac Tromethamine Injection
- Magnesium Sulfate Injection 50%
- Lornoxicam Injection
- Nicardipine HCL Injection
- Diltiazem Injection
- Carboprost Tromethamine 250mcg/1ml
- PROCHLORPERAZINE MESYLATE INJECTION
- AMIKACIN 500mg INJECTION
- Sodium Stibogluconate
- Sodium Hyaluronate Opthalmic Solution 14 mg
- Piroxicam Injection
- Sodium Bicarbonate Injection
- PREDNISOLONE SODIUM PHOSPHATE INJECTION
- Nadroparine Calcium Injection
- Palonosetron Hydrochloride Injection
- NITROGLYCERIN INJCTION USP
- Nefopam Hydrochloride Injection 20mg
- Rabeprazole Sodium Injection
- Quinine Dihydrochloride Injection
- Haloperidol Injection 5mg
- TRIAMCINOLONE ACETONIDE INJECTION
- Lansoprazole Injection
- Neastigmine Methylsulphate Injection
- L-ALANYL-L-GLUTAMINE SOLUTION FOR INFUSION
- VASOPRESSIN INJECTION
- PARECOXIB SODIUM 40 mg injection
- Protamine Sulphate Injection
- Isepamicin Sulfate Injection
- Betamethasone 4mg + Betamethasone 10mg Injection 2ml
- CARBETOCIN INJECTION
- Heparin Injection 5000IU/5ml
- ASCORBIC ACID FOR INJECTION
- Phytomenadion Injection 10mg/ML
- AMOXYCILLIN POTASSIUM
- VITAMIN C 500 INJECTION
- Levomepromazine Hydrochloride Injection
- ETOMIDATE INJECTION 20MG/10ML
- Cyanocobalamin Injection
- BACTERIOSTATIC WATER 0.9% 30ml
- Pentoxifylline Injection
- Ferric Carboxymaltose Injection
- Colecalciferol (Vitamin D3) Injection BP
- Suxamethonium Chloride Injection
- Thiopental Sodium Injection
- Noradrenaline Tartrate Injection
- Bromhexine Hydrochloride Injection 4mg/2ml
- B-Complex With Vitamin B12 Injection
- Ranitidine Injection
- Ampicillin Injection 500
- Aminophylline Injection BP
- Chlorphenamine Injection
- Dextrose Injection USP
- Dobutamine Injection USP
- Gentamicin Injection 20mg
- FLUPENTIXOL INJECTION
- Glycopyrrolate 0.5 Mg Neostigmine Methylsulfate Injection 2.5mg
- Glatiramer Acetate Injection 20 mgml
- LEVETIRACETAM INJECTION 100MG/ML
- Benztropine Mesylate Injection
- Dextrose Injection
- Cloxacillin Injection 1gm
- ISOXSUPRINE HYDROCHLORIDE INJECTION
- L-Carnitine HCL 2000 mg Injection
- NORTHISTERONE ENATHATE INJECTION
- Hydralazine Injection
- Labetalol Hydrochloride Injection
- Cholecalciferol 300000 iu/ml
- Heparin Sodium 25000iu/5ml
- Acetylcysteine Injection 400mg/2ml
- Pyridoxine Hydrochloride Injection 100mg/2ml
- Propranolol Hydrochloride Injection
- Clindamycin Injection 300mg /2ml
- Ergocalciferol Injection 600000/1.5ml
- Fluphenazine Decanoate Injection BP
- Dexamethasone Phosphate Injection 8mg /2ml
- Phenytoin injection 100mg/2ml
- Sodium hyaluronate Opthalmic Solution 18mg PFS
- Trace Elements Injection 3ml
- Dexamethsone sodium injection 8mg /2ml Vial
- Milrinone Lacatate Injection
- Triamcinolone Acetonide Injection Usp
- Tranexamic Acid Injection
- Sodium Chloride Injection
- Flumazenil Injection 0.5mg /5ml
- Phenytoin Sodium Injection 250mg/5ml
- Enoxaparin Sodium Injection 40mg/0.4ml
- Dexmedetomidine hydrochloride injection 50mcg/0.5ml
- Ondansetron Hydrochloride Injection 4mg/2ml
- Esomeprazole Injection
- Medroxyrpogesterone Acetate Injection Suspension Usp
- Ketoprofen Injection
- Promethazine Hydrochloride Injection 50MG/2ML
- METOPROLOL TARTRATE INJECTION
- Alprostadil injection 500mcg/ml
- Pancuronium Injection Bp
- Levosulpiride Injection 25 mg/2ml
- Paricalcitol Injection
- Methocarbamol Injection 1g
- Iron Sucrose Injection
- PRALIDOXIME IODINE INJECTION 500MG/2ML
- GADOBUTROL INJECTION
- Cefoperazone Sulbactams For Injection
- Vitamin b1,b6,b12
- Amikacin Sulfate Injection
- Adrenaline Injection
- Levosimendan Injection
- Sodium Hyaluronate Sterile Solution
- Aminocaproic acid injection 250mg/ml
- Citicoline Injection
- Iopamidol Injection 370
- Gadodiamide Injection 287mg/ml
- Mangnesium Sulfate Injection
- Bacteriostatic 0.9% Sodium Chloride Injection USP 30ML
- AMIODARONE STERILE CONCENTRATE 150M3G 3ML
- Sodium Hyaluronate Sterile Solution 60mg
- Ethylmethlhydroxypryridine Succinate Injection
- Phentolamine Mesylate Bp
- Fluothane Injection
- Iohexol Injection 350 100ml
- Isoflurane USP
- Buserelin Inection
- Diclofenac and Betamethasone and vit.b12 Injection
- Anti-human Thymocyte Immunoglobulin
- 100ml Isoflurane
- Rocuronium bromide injection 50mg/5ml
- Pentoxifylline Injection
- Nefopam Hydrochloride Injection
- Human Plasma Coagulation Factor Viii
- Human Normal Immunoglobulin 5%
- fluorescein 10% 5ml Injection
- Phytomenadione Injection 1mg
- Desflurane Injection
- 250ml Sevoflurane
- Cardioplegia Solution
- Flucloxacillin Injection
- Acetylcysteine Injection 2g /10ml
- iohexol injection
- Amoxicillin And Clavulanate Potassium
- Midacen 500mg Inj.
- Propofol Ocean
- Midazolam 50mg / 10ml Injection
- Metamizole 2.5g 5ml Injection
- Ibuprofen Injection
- HYALURONIC ACID PFS
- Iopromide Injection 300
- Iohexol 300 Injection 100ml
- Hyoscine Butyl Bromide Injection
- Meloxicam Injection
- Gentamicin Injection
- Caffeine Citrate Injection
- Metimizole Sodium Injection
- Norepinephrine Bitartrate injection
- PENTOXYFYLINE INJECTION
- Ambroxol hydrochloride injection
- Thiamine Hydrochloride Injection
- Prochlorperazine Maleate Injection 12.5mg/ml
- Essential phospholipids injection 250mg
- Phenylephrine Hydrochloride
- Chlorpromazine Injection
- Isoprenaline Injection Usp
- Methyprednisolone acetate injection 40mg/ml
- Cisatracurium Besylate Injection 2mg
- Posaconazole Injection 300mg/16.7ml
- Atosiban Acetate Concentrate For Solution For Infusion
- Aprotinin Injection
- Bupivacaine Hydrochloride 5mg In Dextrose 80mg Injection
- Metoclopramide Injection
- Phenylephrine Hydrochlloride Injection Usp
- Piroxicam injection BP 20mg
- Propranolol Hydrochlorie Injection Usp
- Sodium Hyaluronate 3 W-V With Sodium Chondrotin Sulfate
- Calcium Gluconate Injection
- SPECIALITY ANTICANCER MEDICINE
- Pharmaceutical products-1
- Silfeel 50
- Pirfenidone Tablets 200mg
- Vancomycin Intravenous Infusion
- CARBACHOL INTRAOCULAR SOLUTION 0 01 W V
- HYDROCORTISONE TABLETS USP
- UBIQUINOL ACETATE CAPSULES 100MG
- CITICOLINE TABLETS IP 500MG
- PARACETAMOL TABLETS 650MG
- ACETYLCYSTEINE TABLETS 600MG
- POSACONAZOLE ORAL SUSPENSION
- ISOTRETINOIN CAPSULES 20MG
- METHOXSALEN TABLETS
- DECA-PEPTIDE SOLUTION 5ML
- Potassium Chloride Tablet
- ISOTRETINOIN CAPSULES 10MG
- NICORANDIL TABLETS
- Fludrocortisone Tablets
- METOLAZONE TABLETS
- DEXAMETHASONE TABLETS
- Finasteride 5mg Tablet
- SULTAMICILLIN TABLETS 750MG
- TRETINOIN GEL 0.025% W/W
- THYROXINE SODIUM TABLETS
- BENYDAMINE MOUTH WASH B.P.
- ITRACONAZOLE CAPSULES 200MG
- METHYLENE BLUE INJECTION 10MG
- PRUCALOPRIDE TABLETS 1MG
- RIVAROXABAN TABLETS 15MG
- AMANTADINE HYDROCHLORIDE TABLETS 100MG
- CHOLESTYRAMINE FOR ORAL SUSPENSION
- FLUCONAZOLE TABLETS
- CHLOROQUINE PHOSPHATE TABLETS 250MG
- THIAMINE HYDROCHLORIDE, RIBOFLAVIN & PYRIDOXINE HYDROCHLORIDE INJECTION + ASCORBIC ACID, NICOTINAMIDE & ANHYDROUS DEXTROS INJECTION
- BUDESONIDE FORMOTEROL FUMARATE INHALER
- Cilnidipine Tablets
- LEVETIRACETAM TABLETS 500MG
- NIFEDIPINE EXTENDED RELEASE TABLETS 30MG
- Entecavir Tablets 0.5mg
- Paracetamol with ascorbic acid Sachets
- Paracetamol Tablet
- PARACETAMOL TABLET 650MG
- DILOXANIDE FUROATE TABLETS 500MG
- FEBUXOSTAT 80MG TABLET
- Valacyclovir Tablet 500mg
- CINACALCET 30MG TABLET
- Simethicone and Alverine Citrate soft gel capsule
- Entecavir Tablets 1mg
- Folliserum 60ml
- FLIBANSERIN TABLETS 100MG
- Trustive 300mg 600mg 200mg
- Posaconazole Gastro-resistant Tablets 100mg
- Rifaximin Tablets 550mg
- Cefuroxime Axetil Tablet
- Ticarcillin & Clavulanic Acid For Injection
- Rifabutin Capsules
- Micafungin Sodium For Injection 50mg
- Dapagliflozin Tablets
- Nintedanib Soft Gelatin Capsules 150mg
- Estradiol Tablets
- Rizatriptan Tablets 10mg
- Penicillamine Capsules 250mg
- Thyroxine Sodium Tablets 100mcg
- Pancreatin Capsules
- Glycopyrrolate Tablets 2mg
- Nitazoxanide For Oral Suspension
- Bosentan Tablets 62.5mg
- Macitentan Tablets 10mg
- Amiodarone Tablets 200mg
- Desmopressin Acetate Tablets
- Faropenem Tablets 200mg
- Mirabegron Extended Release Tablets 25mg
- PIRFENIDONE TABLETS 801MG
- Ambrisentan Tablets 5mg
- Spironolactone Tablets
- Pentoxifylline Tabelts 400mg
- Carbimazole Tablets
- Cefpodoxime Proxetil Tablets 200mg
- Methimazole Tablets
- Warfarin Sodium Tablet 1mg
- VACCINES
- COMBIPACK OF SCORPION VENOM ANTISERUM WITH STERILE WATER FOR INJECTION
- Rabies Vaccine
- RABIES ANTISERUM EQUINE
- Abhayrab Rabies Vaccine
- HUMAN RABIS IMMUNOGLOBULIN
- DIPHTHERIA AND TETANUS VACCINE FOR ADULTS AND ADOLECENTS
- Hepatitis B Immunoglobulin Human 0.5ml
- HUMAN RABIES IMMUNOGLOBULIN
- VARICELLA VACCINE LIVE
- Hepatitis B Vaccine
- MENINGOCOCCAL POLYSACCHARIDE VACCINE
- HEPATITIS B VACCINE (RDNA)
- HUMAN HEPATITIS B IMMUNOGLOBULINS
- HUMAN HEPATITIS B IMMUNOGLOBULIN 0.5ML
- Tetanus Vaccine (Adsorbed)
- Hepatitis A Vaccine
- PHARMACEUTICAL OINMENTS AND CREAM
- Luliconazole Cream
- Betamethasone Dipropionate Cream
- TRETINOIN CREAM 0.05%
- Ketoconazole Cream
- Clindamycin Phosphate Cream
- GABAPENTIN & DICLOFENAC & METHYL SALICYLATE OINTMENT
- HYDROQUINONE, TRETINOIN & MOMETASONE FUROATE CREAM
- NEOMYCIN SULFATE & FLUOCINOLONE ACETONIDE CREAM
- Tacrolimus Ointment
- Permethrin Lotion
- Permethrin Cream
- Miconazole Nitrate Cream
- PERMETHRIN CREAM
- HYDROQUINONE, TRETINOIN AND FLUOCINOLONE ACETONIDE CREAM
- AMORPHOUS HYDROGEL WOUND DRESSING WITH COLLOIDAL SILVER
- HYDROQUINONE & TRETINOIN & FLUOCINOIONE ACETONIDE TROPICAL CREAM
- Hydrocortisone Acetate Cream
- Diltiazem Gel 2%
- METHOXSALEN SOLUTION
- MICONAZOLE NITRATE NEOMYCIN SULPHATE CLOBETASOL PROPIONATE CREAM
- NADIFLOXACIN CREAM 1 W/W
- SALICYLIC ACID OINTMENT
- DILTIAZEM HYDROCHLORIDE GEL
- Crotamiton Cream
- Benzoyl Peroxide Gel
- Gentamicin Sulfate Cream
- Mupirocin Cream
- FUSIDIC ACID CREAM
- FLUOROURACIL CREAM USP
- Pimecrolimus Cream
- Ivermectin Cream
- DAPSONE GEL 5% W/W
- Phenytoin Sodium 1% Cream
- Clindamycin Phosphate And Benzoyl Peroxide Topical Gel
- NITROGLYCERIN OINTMENT
- MOUTH ULCER GEL
- Hydroxy Methylcellulose Ophthalmic solution USP 2%
- TRETINOIN GEL 0.1% W/W
- XOLABIN INJECTION
- Clindamycin & Nicotinamide Gel
- L-arginine Gel
- Eflornithine Hydrochloride Cream 13.9% W W
- Fluocinolone Acetonide Cream
- Tacrolimus Ointment 0.03% W/w
- Azelaic Acid Cream
- TRETINOIN GEL 0.05% W/W
- Hydroxypropyl Methylcellulose Ophthalmic Gel
- PHARMACEUTICAL GENERAL MEDICINES
- Cloxacillin sodium 500mg Capsule
- 250mg Trientine Hydrochloride Capsules
- Cephalexin 500mg Capsule
- Clotrimazole and Clindamycin Soft gelatin 300mg Capsule
- Benzonatate Capsule
- Itraconazole 100mg Capsule
- Pantoprazole And Itopride Hydrochloride Capsules
- Rifampicin 300mg Capsule
- AMOXICILLIN TRIHYDRATE CAPSULES
- Prostimax Capsules
- AZITHROMCIN CAPSULES 500mg
- Acitretin 25mg Capsules
- AMPICLILING and DICLOXACLILING 500MG
- Fexofenadine Hydrochloride Tablets
- Lansoprazole 30mg Capsule
- Omeprazole 20mg Capsule
- Celecoxib Capsules 200 mg
- Ampicillin 500mg Capsule
- Ampicillin and Dicloxacillin 500mg Capsule
- AMPICLIIN and CLOXACLILING CAPSULE 500MG
- Clindamycin Capsule
- Ulipristal Acetate Capsules 5mg
- Indomethacin 25mg Capsules
- Fenofibrate Capsules 200MG
- PAHRMACEUTICAL PRODUCTS
- Levothyroxine Sodium Tablets
- Potassium Chloride 600mg Tablet
- AZATHIOPRINE TABLETS 50MG
- BETAMETHASONE TABLETS
- Acyclovir tablets 200mg
- Chloroquine Phosphate 250mg Tablet
- Azithromycin Tablets
- Aciclovir 400mg Tablets
- Glyceryl Trinitrate Sublingual Tablet 0.5mg
- Clopidogrel 75mg Tablet
- Quinine Sulphate 100mg Tablet
- Dutas-teride & Tams-ulosin HCL Capsule
- Paracetamol 650mg Tablets
- Mebeverine Hcl 135 Mg,Simethicone 180 Mg And Sulpiride 25 Mg Tablet
- Ivermectin 12mg tablets
- DICYCLOMINE + MEFENAMIC + PARACETAMOL TABLETS
- Allopurinol 300mg Tablets
- Citicoline Sodium 500mg Tablet
- Hydralazine Hcl 25mg Tablet
- Methyldopa Tablets 250mg
- CLOMIPRAMINE HYDROCHLORIDE TABLETS
- ACECLOFENAC TABLETS
- Levothyroxin Sodium 100mcg Tablet
- Salbutamol Sulphate 2mg Tablet
- Ciprofloxacin and Ornidazole Tablet
- Torsemide Tablet
- CLONIDINE 0.150 MG TABLET
- Propranolol Hydrochloride Tablets 40mg
- Pain-Free Ointment
- Prazosin Extended Release Tablets
- Cefixime 200mg Tablet
- Nicorandil 10mg Tablet
- lbuprofen Tablets 400mg
- Montelukast Sodium 10mg Tablet
- Azathioprine 50mg Tablet
- Thiamine Hydrochloride100mg
- Roxithromycin 150mg Tablet
- Mycophenolate Sodium Tablets 500mg
- Moxifloxacin HCL 400mg Tablet
- Paracetamol Effervescent Tablets 650mg
- Allopurinol 100mg Tablets
- Sevelamer 800mg Tablet
- Linezolid 600mg Tablets
- Metoprolol Tartrate 100mg Tablet
- Lorcaserin HCL 10mg Tablet
- Rifampicin Isoniazid Tablet
- Losartan Potassuum 50 mg Tablets
- Prednisolone Tablets
- Clarithromycin 500mg Tablet
- Bismuth Subcitrate Potassium 140mg, Metronidazole 125mg, Tetracycline 125mg Capsules
- Thiamine Tablets
- Verapamil Tablets
- Acetaminophen Tablets 500mg
- Amlodipine Besylate 10mg Tablets
- Artemether AND Lumefantrine 140mg Tablet
- Atorvastatin Tablets 10 mg
- Bisoprolol Fumarate 2.5mg Tablet
- Labetalol 100mg Tablet
- FLAVOXATE TABLETS 200MG
- Folic Acid 5mg Tablets
- Sotalol Tablet 80mg
- Tolperisone HCL 150mg Tablet
- Acetaminophen, Phenylephrine hcl, Dexomethorphen soft gelatin
- Megastrol Tablets IP 40 mg
- Ivermectine 6mg Tablet
- Promethazine Tablet
- Hydroxychloroquine Sulfate 200mg Tablet
- Megastrol Tablets IP 160 mg
- Etoricoxib 60mg Tablet
- Misoprostol 25mcg Tablet
- Ferrous Sulphate 200mg Tablet
- Ondansetron 4mg Tablet
- TOFACITINIB TABLETS 5MG
- Metronidazole 500mg Tablet
- Ondansetron Oral Solution
- Remogliflozin Etabonate Tablet
- Pyridostigmine Bromide 60mg Tablet
- Ondansetron HCL 8mg Tablet
- Alendronate Monosodium 70mg Tablets
- Albendazole 400mg Tablet
- Biperiden 2mg Tablets
- Levocetrizine Diyhydrochloride 5mg Tablet
- Lisinopril And Hydrochlorothiazide Tablet
- Loratadine 10mg Tablet
- Simethicone And Activated Charcoal Tablet
- Sacubitril And Valsartan 50mg Tablet
- Levofloxacin 500mg Tablet
- Isosorbide Dlnltrate 10mg Tablet
- Miso-prostol 25mcg Tablets
- Rabeprazole 20mg Tablet
- Vard-enafil HCL Tablet 20MG
- Zinc Acetate Oral Solution
- Pantoprazole 40mg Tablets
- Azithromycin 250 mg Tablets
- Tamsulosin HCL Capsule
- Sacubitril And Valsartan 100mg Tablet
- Lithium Carbonate 300mg Tablet
- Cetirizine HCL 5mg Tablet
- Ciprofloxacin 500mg Tablet
- Anagrelide Tablets 1
- Meprobamate 400 mg Tablet
- Potassium lodide 100 mcg Tablets
- Co-trimoxazole 960mg Tablet
- Griseofulvin 250mg Tablet
- IBUPROFEN TABLETS BP 600 MG
- Imipramine Hcl 25mg Tablet
- Finasteride Tablet
- Anagrelide Tablets
- Hydralazine Hcl 50mg Tablet
- Pregabalin 300mg Capsule
- Phenazopyridine HCL 100mg Tablet
- Paracetamol And Caffeine Effervescent Tablets
- Clotrimazole Veginal 100mg Tablets
- Voriconazole Tablets 200 mg
- Metronidazole 250mg Tablet
- Nefopam And Paracetamol Tablets
- Hydrochlorothiazide 25mg Tablets
- Levothyroxin Sodium 500mcg Tablet
- Paracetamol, Phenylepherine HCL , Cetrizine HCL And Caffeine Tablet
- Phenytoin Sodium Tablets 25mg
- Piogtitazone HCL 15mg Tablet
- Mefenamic Acid 500mg Tablet
- ONCOLOGY MEDICINES
- PHARMACEUTICAL DRY POWDER AND LYOPHILIZED INJECTION
- Cefepime Injection 1g
- Lomoxicam 8mg Injection
- CEFTRIAXONE FOR INJECTION USP 1G
- Polymyxin B For Injection Usp
- CEFTAZIDIME INJECTION IP
- CEFTAZIDIME FOR INJECTION 1G
- Doxycycline For Injection
- Aztreonam For Injections
- Aciclovir Intravenous Infusion
- Ertapenem For Injection
- Meropenem Injection
- BENZATHINE PENICILLIN INJECTION
- Methylprednisolone Sodium Succinate For Injection
- CEFEPIME TAZOBACTAM FOR INJECTION
- CASPOFUNGIN 70MG Injection
- CEFOTAXIME FOR INJECTION
- Glutathione For Injection 600mg
- Aztreonam For Injection
- Artesunate Injection 120mg
- Indomethacin Injection 1mg
- Tigecycline For Injection 50mg
- Hydrocortisone Sodium Succinate Injection 100
- Liposomal Amphotericin B injection
- CASPOFUNGIN 50MG Injection
- Ceftazidime and avibactam infusion
- Cefepime And Sulbactam Injection
- Fosfomycin For Injection 4g
- Vancomycin Hydrochloride For Intravenous Infusion 1000mg
- Piperacillin Tazobactams Injection
- Colistimethate Sodium For Injection
- SPECIALITY INJECTION
- HORMONAL MEDICINES
- Allylestrenol Tablet 5mg
- Cyproterone acetate 2mg and Ethinyl estradiol 0.03mg tablets
- Estradiol Valerate Tablets
- Medroxyprogesterone Acetate Tablet
- Ethinylestradiol 0.02mg + Desogestre 0.15mg Tablets
- Natural Micronized Progesterone Soft Gelatin 400mg Capsule
- Human Chorionic Gonadotropin (HCG)
- Cabergolin Tablet 1mg
- HUMAN CHORIONIC GONADOTROPHIN 5000IU
- levonorgrel Tablets BP 1.5MG
- Estradiol injection 10mg/1ml
- Recombinate huaman growth hormone 10IU
- Urofollitropin Injection 75IU
- Menotrophin-75IU Injection
- Cetrolix Acetate Injection 0.25 Mg
- Hydroxyprogesterone caproate injection 250mg
- Natural Micronised Progesterone sustained release tablets 200mg
- Progesterone gel 8%w/w
- Medroxyprogesterone Injection
- Hydroxyproesterone caproate 500mg injection
- Ethinylestradiol 0.03mg & Drospirenone 3mg tablets
- Levonorgestrel 0.10mg & Ethinylestradiol 0.02mg tablets
- Boldenone Undecylenate Injection 250mg/ml
- Clenbuterol Hydrochloride EP Tablet 40mcg
- Conjugated Estrogens Tablets
- Drostanolone Enanthate Injection
- Drostanolone Propionate Injection 100mg/1ml
- mesterolone tablets
- Stanozolol Tablets
- Oxandrolone Tablets
- TESTOSTERONE UNDECANOATE SOFTGEL CAPSULE
- Methandienone Tablets
- Dydrogesterone Tablets
- Methenolone Enanthate Injection 10mg /1ml
- Nandrolone Decanoate Injection
- Stanozolol Injection
- testosterone enanthate injection
- Testosterone Propionate Injection
- Testosterone Suspension
- Trenbolone Hexahydro-benzyl-carbonate injection 76.5mg/ml
- Trenbolone Mix Compound Injection 250mg/ml
- PHARMACEUTICAL INJECTION OCEAN
- Pharmaceutical Injection
- Pharmaceutical Products
- HIV AND Hepatitis Medicine
- Atazanvir Ritonavir Tablets
- Efavirenz Tablets 600mg
- Ritovir Tablet
- Darunavir Tablets 600mg
- Zidovudine Tablets 300mg
- Sofosbuvir Dactatasvir Tablets
- Tenofovir Alafenamide Fumarate Tablets 25mg
- Emtricitabine 200mg Tenofovir Alafenamide 25mg Tablets
- Tenofovir Emtricitabine Tablets
- Tenofovir Disoproxil Fumarate & Emtricitabine Tablets
- Combikit Of Lamivudine And Tenofovir Tablets Atazaavir And Ritonavir Tablets
- Emtricitabine And Tenofovir Alafenamide Tablets
- Dolutegravir Emtricitabine And Tenofovir Alafenamide Tablets
- Sofosbuvir 400mg Velpatasvir 100mg Tablet
- Efavirenz Emtricitabine Tenofovir Tablets
- Tenofovir Tablets
- Abacavir Dolutegravir And Lamivudine
- Tenofovir Disoproxil Fumarate And Emtricitabine Tablets
- Sofosbuvir 400mg Velpatasvir 100mg Tablets
- LOPINAVIR 200mg RITONAVIR TABLETS 50mg
- Favipiravir 200mg Tablets
- Dolutegravir Tablets 50mg
- Darunavir & Ritonavir Tablets
- Nevirapine Tablets 200mg
- Tenofovir Disoproxil Fumarate Lamivudine Efavirenz Tablets
- Indinavir Tablets
- Zidovudine Lamivudine Nevirapine Tablets
- Ritonavir Tablets 100mg
- Dolutegravir Emtricitabine Tenofovir Alafenamide Tablets
- Darunavir Ritonavir Tablets
- Dolutegravir, Lamivudine & Tenofovir Disoproxil Fumarate Tablets
- Tenofovir Disoproxil Fumarate Tablets 300mg
- IV Fluids
- Metronidazole Infusion IV
- Moxifloxacin Infusion
- 25D Dextrose Infusion 25%
- NS-Sodium chloride 0.9% infusion
- 10D - Dextrose 10% Infusion
- DNS - Sodium Chloride 0.9% & Dextrose 5% Infusion
- Tinidazole Infusion
- Levofloxacin Infusion
- Ornidazole Infusion
- Pefloxacin infusion
- 25D Dextrose Infusion 25%
- Ofloxacin Infusion
- Ciprofloxacin Infusion
- Fluconazole infusion
- Linezolid infusion
- Mannitol 20% Infusion
- Paracetamol 1g infusion IV
- Anti cancer injection
- Anti cancer medicine
- Nutra and Cosmetics Products
- Inhaler Products
- Contact Us